IlluminOss Photodynamic Bone Stabilization System
EAST PROVIDENCE, R.I., Feb. 6, 2024 /PRNewswire/ — IlluminOss Medical, offering a unique, minimally invasive technology for fracture repair and stabilization in geriatric and compromised bone, announced it has reached the important benchmark of enrolling over 200 patients in its global device registry across 16 sites. IlluminOss will highlight the results of the patients in the registry at the upcoming American Academy of Orthopedic Surgeons meeting.
To further the evidence of the 1000+ patients’ data in peer reviewed publications and conference proceedings, IlluminOss launched a clinical registry to demonstrate the real-world use of our device. The registry is designed to collect safety and performance data on the IlluminOss Device when used to provide stabilization and alignment for the treatment of fractures in compromised bone. The registry aims to show real world outcomes for the system. Standard of care follow up visit data is collected at 75 days, 6 months, and 1 year from the initial procedure. Over half of the patients enrolled have already reached their first follow up visit.
About the IlluminOss Photodynamic Bone Stabilization System
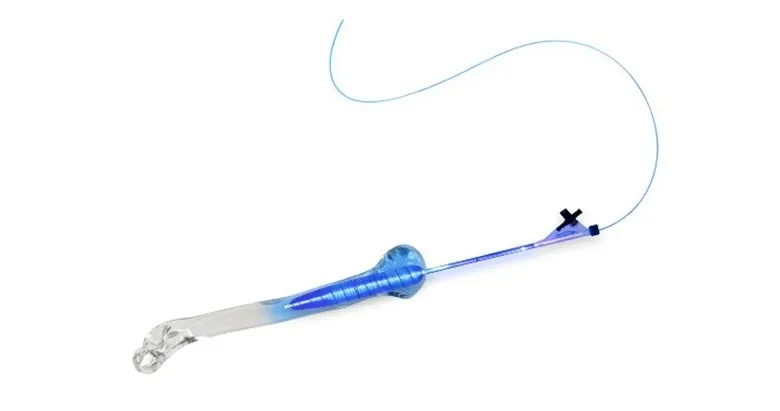
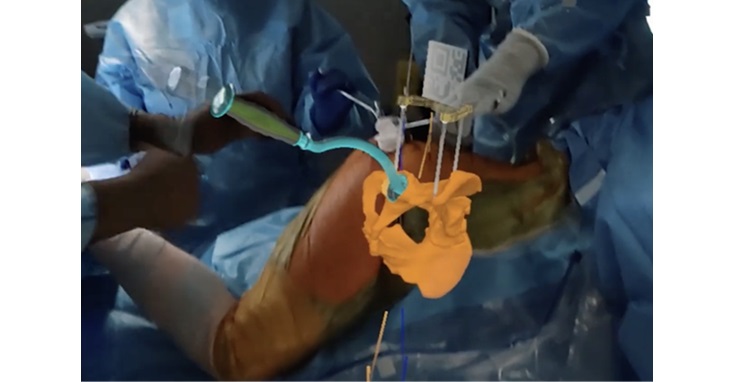
IlluminOss technology involves expanding a balloon-like implant inside a fractured bone by infusing the implant with a light-curing liquid. Blue light is then transmitted to the implant via an optical fiber to convert the liquid into a polymer, creating a strong core within the bone. This stabilizes the fracture and allows for a fast return to daily activities. Some patients are able to regain use of their injured limb the day of surgery. If additional fracture fixation is necessary, the strength supplied by the IlluminOss implant allows surgical plates and screws to be securely fastened into the bone.
Dr. Jason Halvorson, Orthopedic trauma surgeon and study investigator from Raleigh, NC says “I started using this only for periprosthetic fractures and have now used it when I need to increase stability and the construct stiffness of the fracture. I don’t have to worry about screwing into the bone. The IlluminOss system augments poor bone, allowing for an increase in bone strength. Having the IlluminOss implant gives my patients the opportunity for early mobilization and weight bearing.”
About IlluminOss Medical, Inc.
Headquartered in East Providence, RI, IlluminOss is a privately held, commercial-stage medical device company. The IlluminOss system is CE-marked and FDA-cleared for a variety of anatomical sites, with further indications pending. For additional information, including a complete list of indications, contraindications, warnings, precautions and risks, visit www.illuminoss.com.
For any press questions, please contact Lisa Holt, at lholt@illuminoss.com.
SOURCE IlluminOss Medical