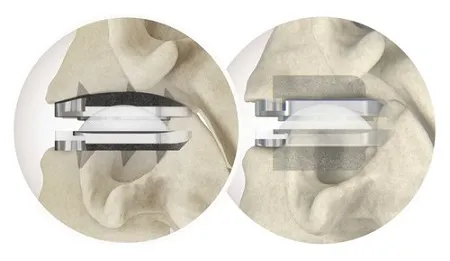
Huntsville, AL based technology company, Curiteva announces the first FDA 510(k) cleared 3D printed PEEK implant, the Inspire Porous PEEK Cervical Interbody System with HAFUSE Technology. The Inspire platform is manufactured with a proprietary, patented Fused Filament Fabrication 3D printer designed, programmed, and built by Curiteva. This ground-breaking additive process produces a fully interconnected and integrated porous structure traversing the entire implant to promote osseointegration, improve radiographic assessment, and deliver superior biomechanics. The first-to-market combination of the HAFUSE nanotechnology surface treatment and novel porous PEEK structure creates a hydrophilic, bioactive environment for cell attachment, proliferation, and healing in pre-clinical animal and in vitro studies.
“The distinctive Inspire implant technology enabled by our innovative 3D printing process incorporates an engineered lattice structure with fully interconnected porosity exhibiting superior mechanical strength and achieving a modulus of elasticity closely matching human cancellous bone,” said Co-Founder and Chief Technology Officer Eric Linder.
“Our capability to integrate unique design elements utilizing additive PEEK as an alternative to titanium to optimize beneficial material properties such as radiographic assessment and biomechanical load-sharing represents a significant leap forward in the design and development of medical devices,” added Inventor and Vice President of Emerging Technology Todd Reith.
The company plans a commercial launch in key academic centers across the United States. “Curiteva is pioneering 3D printing of porous PEEK implants with a bioactive surface to revolutionize how engineered structures and implant biomaterials enhance healing and improve patient outcomes. We are uniquely positioned to control the product development process of traditional implants and 3D printed devices from inception to commercialization and scale to meet market demand inside our 35,000 sq ft production facility in Huntsville,” stated CEO Mike English.
Curiteva is a privately held technology and manufacturing company dedicated to advancing spine surgery and improving clinical outcomes by partnering with providers and suppliers to deliver innovative and intuitive implant systems to the market. Our business is founded on a commitment to building world-class manufacturing, accelerating research and development, maintaining lean operational discipline, and delivering novel technology to meet the evolving needs of our customers and the patients they serve. For more information, please visit www.curiteva.com.